Carbon is naturally in all living organisms and is replenished in the tissues by eating other organisms or by breathing air that contains carbon. At any particular time all living organisms have approximately the same ratio of carbon 12 to carbon 14 in their tissues.
Radiocarbon dating - Wikipedia
When an organism dies it ceases to replenish carbon in its tissues and the decay of carbon 14 to nitrogen 14 changes the ratio of carbon 12 to carbon Experts can compare the ratio of carbon 12 to carbon 14 in dead material to the ratio when the organism was alive to estimate the date of its death. Radiocarbon dating can be used on samples of bone, cloth, wood and plant fibers.
The half-life of a radioactive isotope describes the amount of time that it takes half of the isotope in a sample to decay. In the case of radiocarbon dating, the half-life of carbon 14 is 5, years.
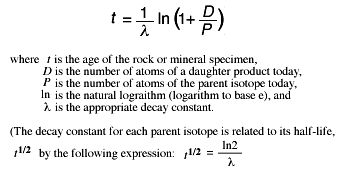
This half life is a relatively small number, which means that carbon 14 dating is not particularly helpful for very recent deaths and deaths more than 50, years ago. After 5, years, the amount of carbon 14 left in the body is half of the original amount. Simplifying this expression by canceling the N 0 on both sides of the equation gives,. Solving for the unknown, k , we take the natural logarithm of both sides,.
- Blank corrected fraction modern.
- .
- .
Other radioactive isotopes are also used to date fossils. The half-life for 14 C is approximately years, therefore the 14 C isotope is only useful for dating fossils up to about 50, years old. Fossils older than 50, years may have an undetectable amount of 14 C. For older fossils, an isotope with a longer half-life should be used. For example, the radioactive isotope potassium decays to argon with a half life of 1.
Other isotopes commonly used for dating include uranium half-life of 4.
Radiocarbon dating
Problem 1- Calculate the amount of 14 C remaining in a sample. Problem 2- Calculate the age of a fossil. Problem 3- Calculate the initial amount of 14 C in a fossil.
- is john mayer still dating katy perry.
- What is Radiocarbon Dating?.
- Process Blanks.
Problem 4 - Calculate the age of a fossil. Problem 5- Calculate the amount of 14 C remaining after a given time has passed.
Navigation menu
Decay of radioactive isotopes Radioactive isotopes, such as 14 C, decay exponentially. Modeling the decay of 14 C. Thus, we can write: